Research Topics
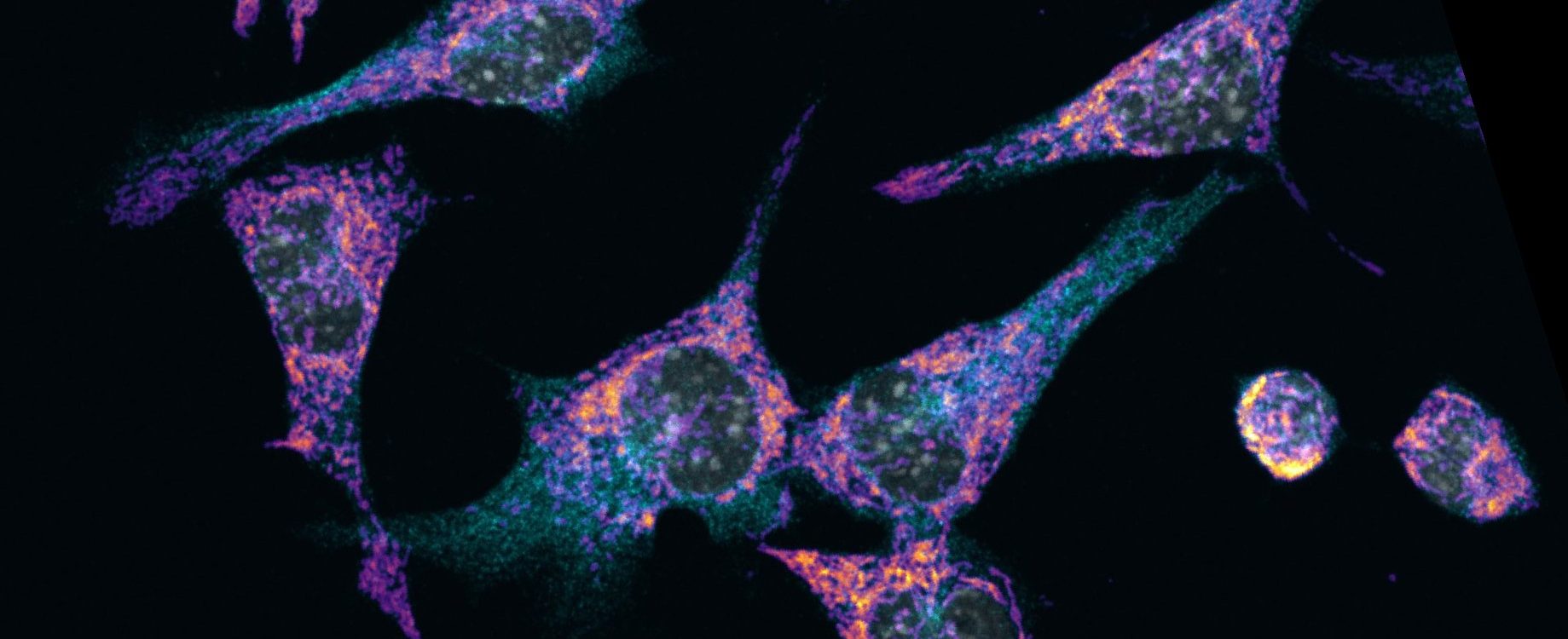
The small GTPase RAB24
We showed that RAB24 is needed at a late stage of the macroautophagy pathway. Several studies show that RAB24 is important for health. It supports the survival of neurons, and mutations in RAB24 cause ataxia, lack of coordination of muscle movements, in dogs. RAB24 promotes malignancy of liver cancer, and its overexpression enhances liver fat accumulation. RAB24 facilitates survival of liver cancer cells by decreasing cell death, promoting growth and increasing cell motility.
 We aim to clarify the biological functions of RAB24, as well as the molecular mechanisms that mediate these functions.We have screened for proteins that interact with RAB24 and are currently investigating the molecular mechanisms they use to regulate RAB24. Our research on the molecular regulation of RAB24 has potential to reveal new insights on RAB proteins. Our results will also help to understand the molecular pathogenesis of human diseases. Our long-term goal is to reveal new targets for development of treatments for neurodegeneration, cancer and fatty liver disease.
We aim to clarify the biological functions of RAB24, as well as the molecular mechanisms that mediate these functions.We have screened for proteins that interact with RAB24 and are currently investigating the molecular mechanisms they use to regulate RAB24. Our research on the molecular regulation of RAB24 has potential to reveal new insights on RAB proteins. Our results will also help to understand the molecular pathogenesis of human diseases. Our long-term goal is to reveal new targets for development of treatments for neurodegeneration, cancer and fatty liver disease.
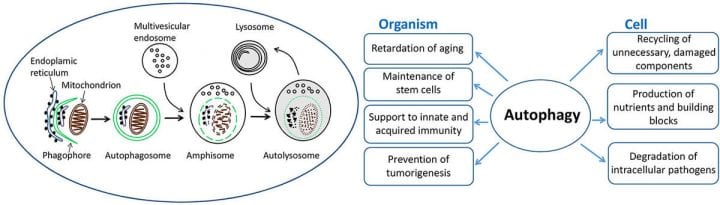
Membrane dynamics during autophagosome biogenesis
Autophagy is a lysosomal degradation pathway for cytoplasmic material and organelles that maintains cellular homeostasis during nutrient deprivation and stress. Autophagy is an important survival mechanism during short-term starvation: by degrading non-essential components, cells acquire nutrients for energy production and vital biosynthetic reactions. Autophagy also contributes to the clearance of damaged organelles and aggregate-prone proteins, protection against metabolic stress and DNA damage, and even to longevity of the organism. Defective autophagy has been connected to many human diseases including cancer, myopathies, metabolic diseases, and neurodegenerative diseases.
We and others showed that autophagosomes form in a subcompartment of the endoplasmic reticulum. A flat membrane cistern called the phagophore emerges first, which then elongates and forms a double-membrane bound autophagosome. Autophagosomes acquire hydrolytic capacity by fusing with endosomes and lysosomes. The origin of phagophore and autophagosome membranes is one of the longest lasting open questions in the field. We investigate the membrane dynamics and organelle contacts during phagophore biogenesis using three-dimensional electron microscopy, live-cell imaging and correlative light-electron microscopy.
Further reading on RAB24:
Ylä-Anttila P, Mikkonen E, Happonen KE, Holland P, Ueno T, Simonsen A, Eskelinen E-L: RAB24 facilitates clearance of autophagic compartments during basal conditions. Autophagy 2015, 11:1833-48. DOI: 10.1080/15548627.2015.1086522
Ylä-Anttila P, Eskelinen E-L: Roles for RAB24 in autophagy and disease. Review. Small GTPases 2018, 9:57-65. DOI: 10.1080/21541248.2017.1317699
Further reading on autophagy:
Klionsky DJ, Petroni G, Amaravadi RK, Baehrecke EH, Ballabio A, Boya P, Bravo-San Pedro JM, Cadwell K, Cecconi F, Choi AMK, Choi ME, Chu CT, Codogno P, Colombo MI, Cuervo AM, Deretic V, Dikic I, Elazar Z, Eskelinen E-L, Fimia GM, Gewirtz DA, Green DR, Hansen M, Jäättelä M, Johansen T, Juhász G, Karantza V, Kraft C, Kroemer G, Ktistakis NT, Kumar S, Lopez-Otin C, Macleod KF, Madeo F, Martinez J, Meléndez A, Mizushima N, Münz C, Penninger JM, Perera RM, Piacentini M, Reggiori F, Rubinsztein DC, Ryan KM, Sadoshima J, Santambrogio L, Scorrano L, Simon HU, Simon AK, Simonsen A, Stolz A, Tavernarakis N, Tooze SA, Yoshimori T, Yuan J, Yue Z, Zhong Q, Galluzzi L, Pietrocola F: Autophagy in major human diseases. Review. EMBO J 2021, 40:e108863. DOI: 10.15252/embj.2021108863
Eskelinen E-L: Autophagy: supporting cellular and organismal homeostasis by self-eating. Review. Int J Biochem Cell Biol 2019, 111:1-10. DOI: https://doi.org/10.1016/j.biocel.2019.03.010
Recent and present funding
Currently, our work is supported by the University of Turku, Research Council of Finland (formerly the Academy of Finland) and Jane and Aatos Erkko Foundation. The logos below show the funding bodies that have supported our work in Turku since 2017.